The bacteria in your intestines are manufacturing the same chemicals your psychiatrist prescribes, and they have been doing it for millions of years before anyone thought to put serotonin in a pill. That is not a metaphor or a loose analogy. It is a biochemical reality that has fundamentally shifted how researchers think about mood, motivation, and mental resilience in 2026.
For decades, the conversation about neurotransmitters lived entirely inside neuroscience departments. Serotonin was a brain chemical. Dopamine was a brain chemical. GABA was a brain chemical. The gut was just plumbing. But a growing body of evidence, accelerating rapidly over the past five years, has revealed something the medical establishment was slow to accept: your gastrointestinal tract is not a passive bystander in your emotional life. It is a primary player. And the trillions of microorganisms living inside it are running a neurochemical production line that your brain depends on far more than most people realize.
If you have been exploring how the gut-brain connection influences your mental wellness, this is the layer underneath. This is the factory floor.
Your gut is a neurotransmitter factory (and it never clocks out)
The human gut microbiome contains somewhere between 30 and 40 trillion bacterial cells. That number alone is staggering, but what matters more than the sheer count is what those bacteria are doing. Among their many functions, certain species actively synthesize neurotransmitters or their direct precursors. They do this as part of their own metabolic processes, and the byproducts happen to be molecules that your nervous system uses to regulate mood, focus, stress tolerance, and sleep.
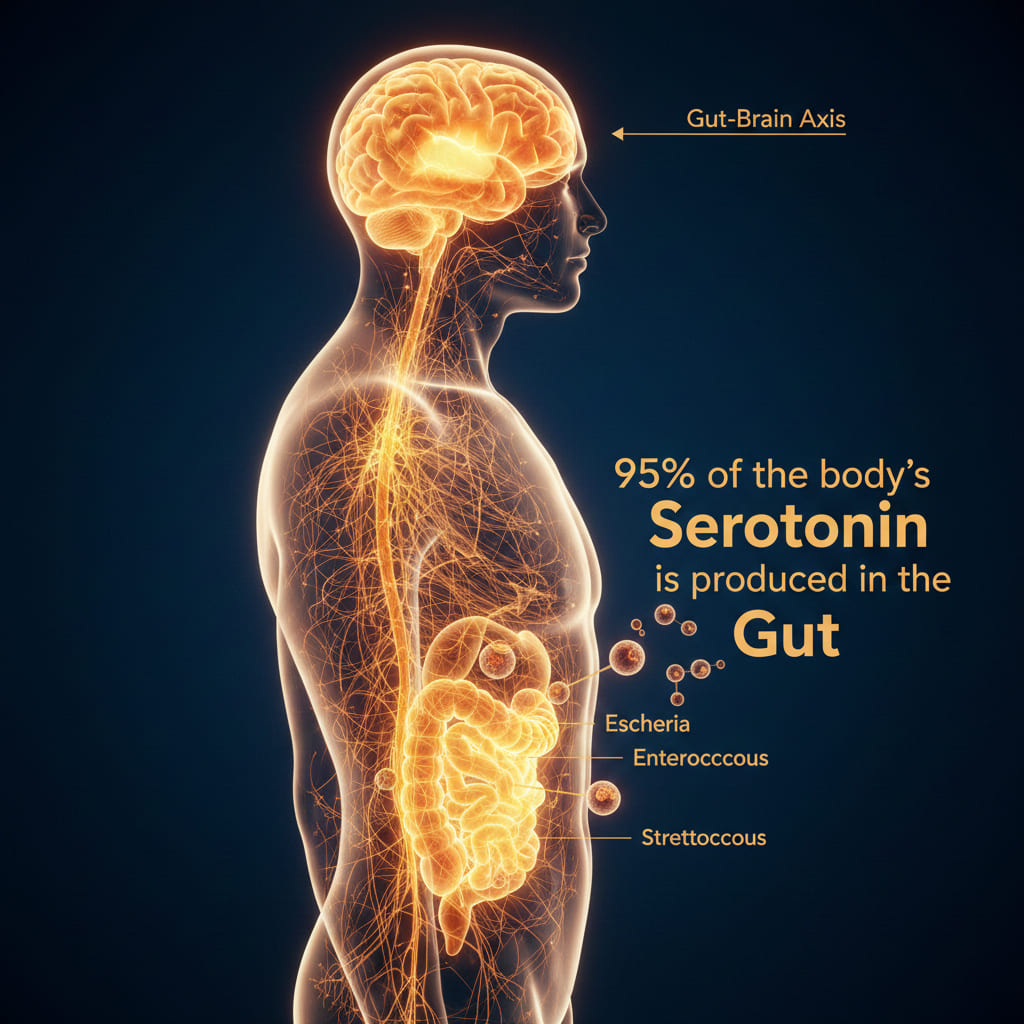
This is not a minor side operation. The gut produces approximately 95% of the body’s total serotonin supply. Let that figure land for a moment. The molecule most commonly associated with happiness, emotional stability, and the mechanism of action behind SSRIs like Prozac and Zoloft is overwhelmingly manufactured outside the brain.
Enterochromaffin cells lining the intestinal wall handle the synthesis, converting the amino acid tryptophan into serotonin. But here is the part that changes the picture: gut bacteria directly influence how much tryptophan is available for that conversion and how efficiently the process runs. Species of Escherichia, Enterococcus, and Streptococcus have all been documented participating in tryptophan metabolism. Remove them, and serotonin output drops measurably.
The GABA story: why your gut bacteria might be better at calming you down than your breathing app
GABA (gamma-aminobutyric acid) is the brain’s primary inhibitory neurotransmitter. When GABA is functioning well, you feel calm, centered, capable of handling stress without spiraling. When GABA signaling is disrupted, anxiety rises, sleep suffers, and that background hum of nervous tension becomes your default state.
What most people do not know is that several common gut bacteria produce GABA directly. Lactobacillus brevis and Bifidobacterium dentium are among the most efficient GABA producers identified so far. In a 2023 study published in Nature Microbiology, researchers mapped GABA production capacity across hundreds of bacterial strains and found that the gut’s ability to generate this calming neurotransmitter varies dramatically from person to person, largely based on microbiome composition.
The implications are significant. Two people can face the same stressful situation and have genuinely different neurochemical responses, not because of willpower or resilience training, but because one person’s gut bacteria are producing more GABA than the other’s.
I have spent years advising clients on nutritional strategies for stress management, and honestly, understanding the GABA-microbiome link changed how I approach those conversations. It reframed anxiety as something partially rooted in ecology, not just psychology.
Dopamine, norepinephrine, and the precursor pipeline
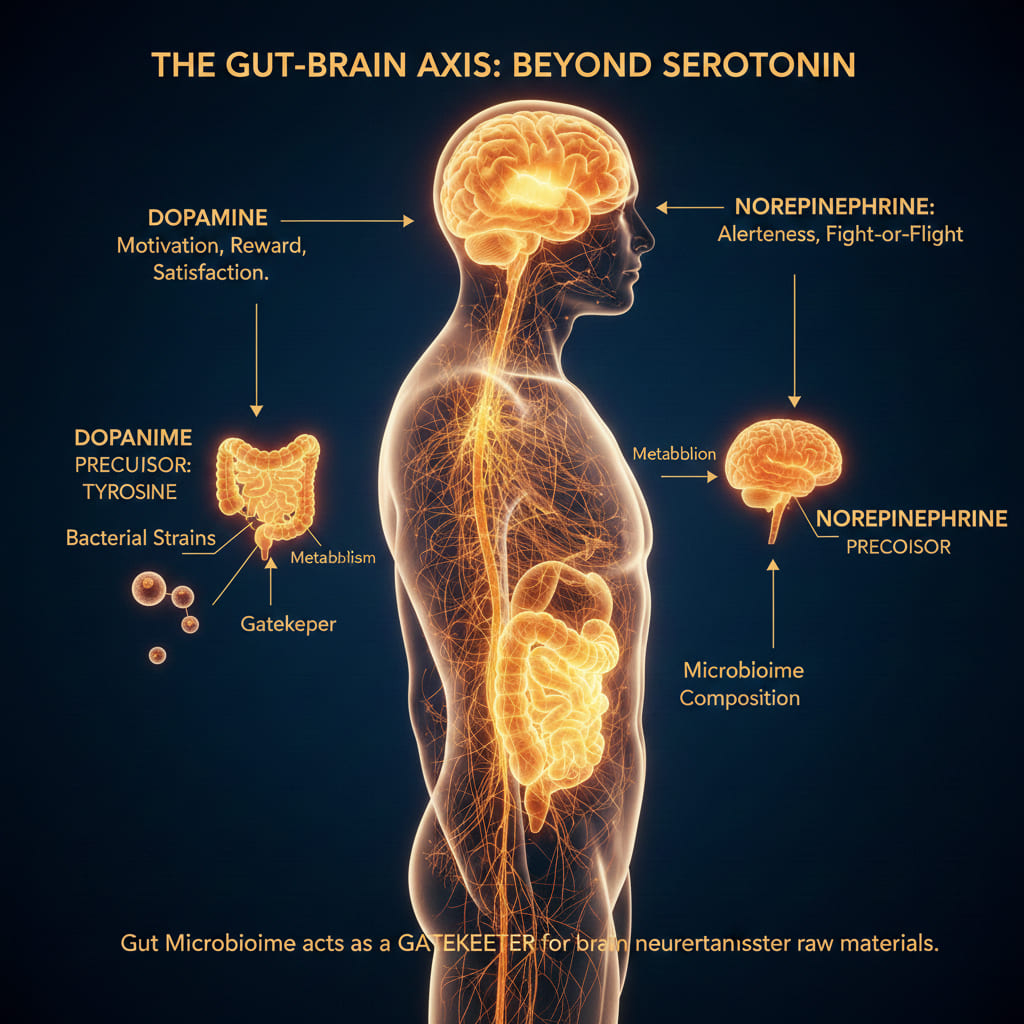
Serotonin and GABA get the most attention in gut-brain research, but the story extends further. Your gut microbiome also influences the availability of dopamine and norepinephrine precursors.
Dopamine drives motivation, reward-seeking behavior, and the feeling of satisfaction when you accomplish something. Its precursor, the amino acid tyrosine, is metabolized by gut bacteria before it ever reaches the brain. Certain bacterial strains enhance tyrosine availability. Others compete for it or convert it into different metabolites entirely.
Norepinephrine, which governs alertness and the fight-or-flight response, follows a similar pattern. Research from University College Cork, published in 2022, demonstrated that germ-free mice (raised without any gut bacteria) had significantly altered norepinephrine levels in the brain compared to conventionally colonized animals. Reintroducing specific bacterial communities normalized those levels.
The practical takeaway: the raw materials your brain uses to build its chemical messengers are being processed, allocated, and sometimes consumed by your gut microbiome before they reach their intended destination. The composition of your microbiome acts as a gatekeeper.
Why microbial diversity is the variable that matters most
If you are reading this and thinking, “So I just need to find the right bacterial strain and take a supplement,” the reality is more nuanced. Individual strains matter, and the emerging field of probiotics for anxiety and depression is producing genuinely exciting clinical results. But the broader principle is about ecosystem diversity.
A microbiome dominated by a narrow range of species tends to produce a narrow range of metabolites. A diverse microbiome, one populated by hundreds of different bacterial species, generates a wider array of neurotransmitter precursors, short-chain fatty acids, and anti-inflammatory compounds. This diversity acts as a buffer against neurochemical imbalance.
The American Gut Project, one of the largest citizen-science microbiome studies ever conducted, found that the single strongest predictor of microbiome diversity was not probiotic use. It was the number of different plant species a person consumed per week. Participants eating 30 or more distinct plants weekly had significantly more diverse microbiomes than those eating 10 or fewer.
That finding aligns with what I tell clients constantly: variety on your plate translates to variety in your gut, which translates to a broader neurochemical toolkit for your brain.
The inflammation bridge: when bad bugs send bad signals
Not all microbial influence on mood is positive. When the microbiome falls out of balance, a state called dysbiosis, the consequences for neurotransmitter production can be severe.
Dysbiosis often involves an overgrowth of pro-inflammatory species and a decline in beneficial ones. The resulting inflammation does not stay local. Pro-inflammatory cytokines produced in the gut enter the bloodstream, cross the blood-brain barrier, and interfere with neurotransmitter metabolism directly. Specifically, inflammation diverts tryptophan away from serotonin production and toward the kynurenine pathway, which generates neurotoxic metabolites instead of mood-stabilizing ones.
This is one reason why chronic digestive issues so frequently co-occur with depression and anxiety. It is not that people with stomachaches are just “stressed.” It is that the inflammatory state in their gut is literally rerouting the biochemistry their brain relies on for emotional regulation.
A 2024 meta-analysis in Translational Psychiatry reviewed 41 studies and confirmed a consistent association between elevated gut-derived inflammatory markers and reduced central serotonin availability. The connection is no longer speculative. It is mechanistic and measurable.
What you can do starting this week
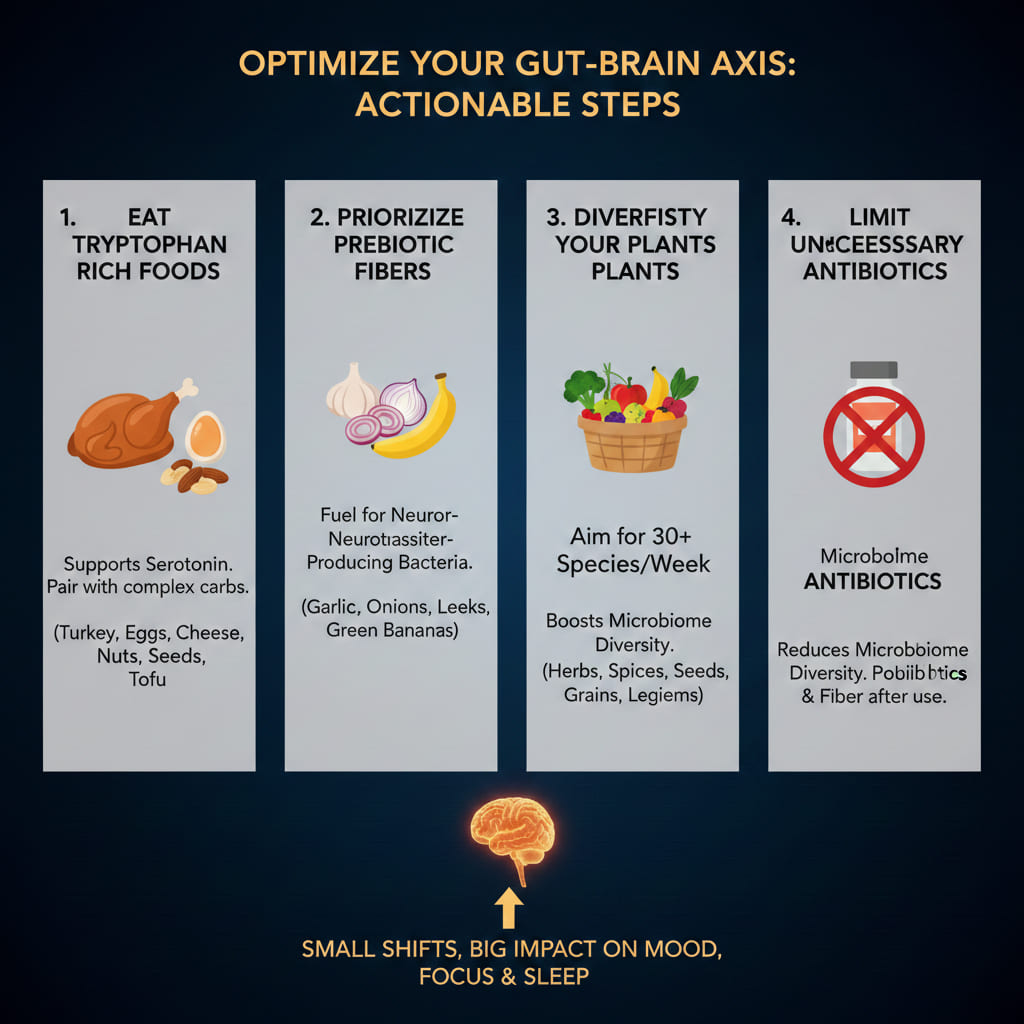
You do not need to sequence your microbiome or overhaul your diet overnight to start supporting neurotransmitter production in your gut. A few targeted shifts make a measurable difference.
Eat more tryptophan-rich foods. Turkey, eggs, cheese, nuts, seeds, and tofu all supply the amino acid your gut converts into serotonin. Pairing these with complex carbohydrates improves tryptophan’s ability to cross the blood-brain barrier.
Prioritize prebiotic fibers. Garlic, onions, leeks, asparagus, and slightly green bananas feed the bacterial species responsible for GABA and SCFA production. These fibers are the fuel your neurotransmitter-producing bacteria run on.
Diversify your plants. Challenge yourself to hit 30 different plant species per week. Herbs and spices count. So do nuts, seeds, grains, legumes, fruits, and vegetables. Each one feeds a slightly different microbial population.
Limit unnecessary antibiotic use. Antibiotics are life-saving medicines when needed, but each course reduces microbiome diversity. If you have recently completed a course, a targeted probiotic protocol and increased fiber intake can help rebuild what was lost.
Where the science goes from here
We are still in the early chapters of understanding exactly how gut bacteria communicate with the brain at a molecular level. But the trajectory is clear. Major pharmaceutical companies are investing in “psychobiotic” drug development. The NIH Human Microbiome Project continues to map the functional capabilities of intestinal bacteria. And nutritional psychiatry, a field that barely existed a decade ago, is now represented at every major psychiatric conference.
The gut is not simply digesting your lunch. It is producing the chemicals that determine whether you feel motivated or defeated, calm or anxious, sharp or foggy. And the most remarkable part is that you have more control over the composition of your gut microbiome, and therefore over this neurochemical production line, than you have over almost any other factor in your mental health.
So here is the question worth sitting with: if you knew that the bacteria in your gut were shaping your emotional baseline every single day, would you feed them any differently tonight?