The bacteria living in your intestines may be silently trading the very genes that make superbugs unstoppable. A comprehensive review published in February 2026 in the British Journal of Biomedical Science reveals that while scientists race to develop new antibiotics and alternative therapies, the gut microbiome has emerged as both the problem and potentially the solution in the global antimicrobial resistance crisis.
The gut as a resistance gene factory
Research now confirms what microbiologists have long suspected: your gastrointestinal tract functions as a reservoir and active exchange hub for antimicrobial resistance genes. The gut microbiome encodes nearly three million genes, vastly outnumbering the roughly 23,000 genes in the human genome, and this genetic diversity includes numerous resistance determinants that can spread between bacterial species through horizontal gene transfer.
The mechanisms driving this spread include conjugation, transduction, and transformation. Essentially, bacteria swap genetic material like trading cards, passing resistance genes from harmless commensals to potentially deadly pathogens. A 2025 Frontiers in Microbiology review described the gut as “an emerging epicenter of antimicrobial resistance,” noting that polymicrobial communities within intestinal biofilms particularly facilitate the development and spread of resistance via horizontal gene transfer.
What makes this concerning is the sheer scale. Without effective interventions, antimicrobial resistance is projected to cause an additional 39 million deaths worldwide between 2025 and 2050, according to WHO projections cited in recent research. The gut, it turns out, is where much of this resistance develops and spreads.
From crisis to toolkit: novel therapeutic approaches
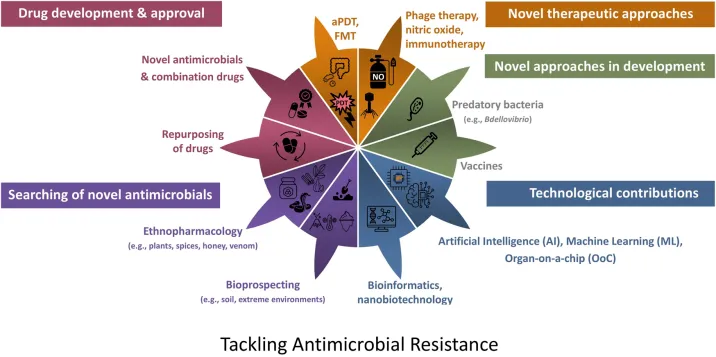
The British Journal of Biomedical Science review catalogues an impressive arsenal of emerging therapies targeting resistant infections. These approaches span from cutting-edge pharmaceuticals to surprisingly ancient remedies.
Microbiome manipulation and fecal transplantation
Fecal microbiota transplantation (FMT) has evolved from a niche therapy into a cornerstone treatment for recurrent Clostridioides difficile infection, achieving sustained response rates surpassing 80% in select populations. The FDA has now approved two standardized microbiota-based therapeutics: Rebyota and Vowst.
More intriguingly, FMT shows promise for decolonizing patients carrying multidrug-resistant organisms. A randomized controlled trial called PREMIX demonstrated that eight of nine patients who completed FMT treatment became culture-negative for multidrug-resistant organisms. The mechanism appears to involve competitive exclusion, where transplanted beneficial bacteria outcompete and displace resistant pathogens.
Phage therapy gains clinical ground
Bacteriophages, viruses that specifically target and destroy bacteria, are advancing through clinical trials for infections ranging from diabetic foot ulcers to cystic fibrosis lung infections. The review documents over 30 ongoing or recently completed clinical trials investigating phage therapy.
The first clinical success came in 2017 when a nine-phage cocktail successfully treated a multidrug-resistant Acinetobacter baumannii infection in a diabetic patient. Since then, the University of California San Diego established the Center for Innovative Phage Applications and Therapeutics (IPATH), reporting that such therapy can function both as treatment and as suppressive therapy for chronic infections.
Natural compounds show surprising potential
Researchers are bioprospecting extreme environments, from Antarctic marine sediments to the Dead Sea, discovering novel antimicrobial compounds. Antarctic fish produce antimicrobial peptides called piscidins with activity against multidrug-resistant bacteria. Dead Sea soil extracts have demonstrated antibiofilm activity against wound pathogens.
Even honey has received renewed scientific attention. Manuka honey contains methylglyoxal and other compounds that disrupt bacterial membranes and biofilms. Clinical trials are currently investigating medical-grade honey for treating deep neck abscesses and burn wounds.
What this means for your gut health
The gut-resistance connection has practical implications. Antibiotic use, even when medically necessary, disrupts the intestinal ecosystem and can promote resistance gene exchange. Research published in Nature Microbiology in December 2025 found that 168 common industrial and agricultural chemicals also damage beneficial gut bacteria, and when stressed by these chemicals, some bacteria may become resistant to antibiotics.
This underscores the importance of supporting gut microbial diversity through diet and lifestyle choices. A diverse, fiber-rich diet feeds beneficial bacteria that provide WHO Bacterial Priority Pathogens List 2024 against pathogens. When harmful bacteria cannot establish themselves, there are fewer opportunities for resistance genes to spread.
Probiotics and fermented foods may offer additional support, though research on their specific role in resistance prevention remains ongoing. The key insight is that gut health and antimicrobial resistance are fundamentally connected.
The bottom line
The antimicrobial resistance crisis demands innovation beyond traditional antibiotics. Scientists are now exploring phage therapy, antimicrobial peptides, microbiome manipulation, and compounds from extreme environments. Two FDA-approved fecal microbiota products are already available, with more in development. But perhaps the most actionable insight is this: protecting your gut microbiome through dietary choices and judicious antibiotic use may help limit the spread of resistance genes in your own body.
The gut that evolved to protect us may also hold keys to protecting our antibiotics.
How concerned are you about antibiotic resistance, and have you made any changes to support your gut health because of it?